Regulatory Affairs
The compliance of your clinical trials ensured by our CRO
Your compliance, ensured by our Regulatory Affairs services.
Our experts secure your interactions with the authorities and support you in managing your clinical trials and regulatory documentation, in compliance with ICH-GCP, ISO 14155 and 21 CFR Part 11 requirements. We take care of your obligations under the MDR 2017/745 and IVDR regulations, including key activities such as the Clinical Evaluation Report (CER) and PMCF for medical devices.
The goal: zero surprises, zero rejections, zero critical deviations.
Our areas of expertise
CRO Regulatory Affairs: comprehensive management of your regulatory obligations
We cover the entire regulatory scope to ensure the compliance of your medical devices and trials:
Regulatory submissions
Clinical Trial Application (CTIS), health authorities, RA/MOH, IRB, Ethics Committee
Clinical & pharmaceutical documentation
IMPD, IB, CEP, PSUR, DSUR files, CER clinical evaluation report, medical device PMCF plans and reports
Thanks to our expertise in regulatory affairs CRO, we ensure the consistency and compliance of your documentation throughout the product life cycle.
Regulatory Affairs
Regulatory affairs management: MDR/IVDR compliance and document control
Centaur Clinical provides you with a team of experts to consolidate your technical file. We ensure that your medical devices comply with Regulation (EU) 2017/745 (MDR) and Regulation (EU) 2017/746 (IVDR), prepare and update your CER, PMCF plans and reports, and coordinate your clinical trials. We guarantee document control (eTMF QC), quality monitoring, CAPA management and preparation for audits and inspections.
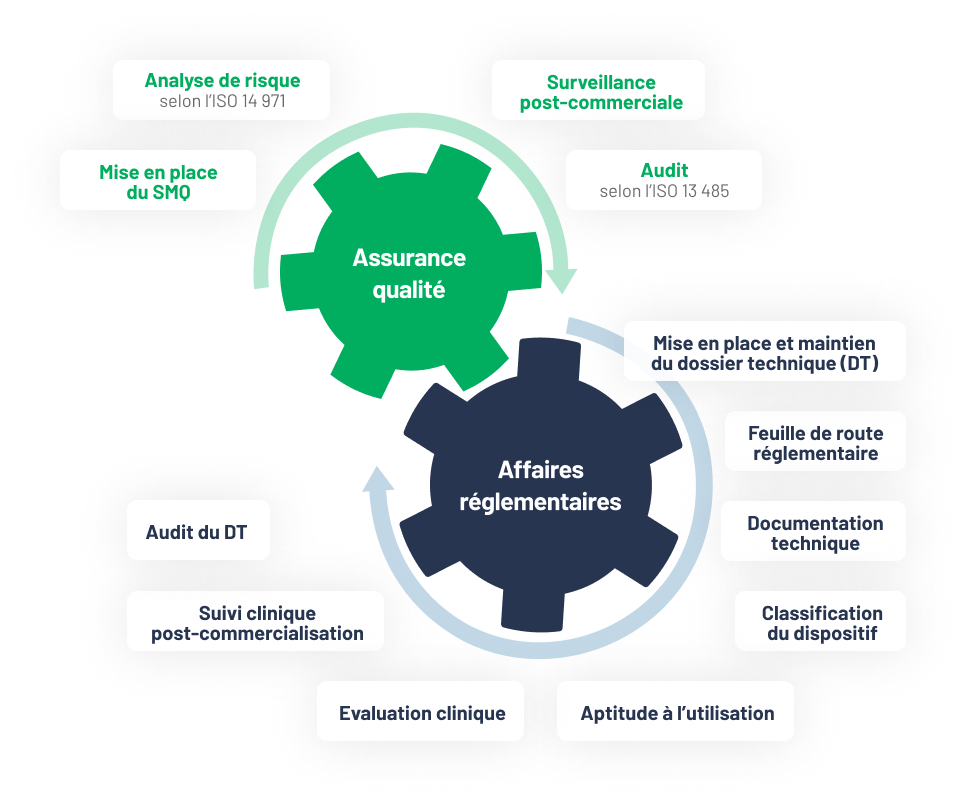
Regulatory analysis and strategy
Identification of applicable requirements (MDR/IVDR, countries), gap analysis, submission plan, clinical strategy (CER, PMCF), and document mapping (IMPD, IB, CEP, PSUR/DSUR).
Submissions and interactions with authorities
Preparation of CTA/CTIS files, coordination with authorities (RA/MOH) and ethics committees, responses to questions (Q&A), variations, document updates until approval.
I have a project
Centaur Clinical supports you at any stage of the Technical Documentation (TD) and the Quality Management System (QMS)
We support manufacturers throughout the CE certification process with the Notified Body.

Testimonials
Our satisfied clients talk about us
We are starting a clinical investigation with Centaur Clinical and are very satisfied with our collaboration so far. Preparing the submission to the authorities is a stressful and time-consuming process that requires qualified personnel. The Centaur Clinical teams are always on hand to provide us with their expertise, availability and support.

Centaur Clinical helped us define a rationale and a clinical investigation strategy that was robust and consistent with the expectations of health authorities regarding the application of the RDM and the clinical evidence required.

